Viruses exacerbate disease caused by Leishmania parasite
Evolution of parasite viruses hints at how Leishmania first infected vertebrates
 Getty Images
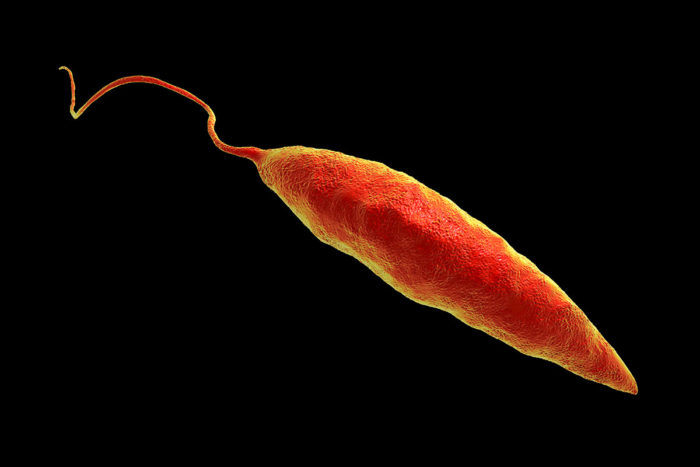
Getty ImagesStephen Beverley, PhD, found that viruses hidden inside the parasite Leishmania (above) worsen disease caused by the parasite. Now, an evolutionary survey of the viruses in related parasites suggests that Leishmania’s viruses may have helped it make the jump from infecting insects to infecting vertebrates.
More than a million people in tropical countries contract the parasite Leishmania every year through the bites of infected sand flies. Most people develop disfiguring – but not life-threatening – skin lesions at the sites of the bites. But if the parasite spreads to the internal organs, it causes a disease known as visceral leishmaniasis, which kills about 30,000 people every year.
Stephen Beverley, PhD, the Marvin A. Brennecke Professor and head of the Department of Molecular Microbiology at Washington University School of Medicine in St. Louis, is a world expert on the deadly parasite. He was studying Leishmania’s basic biology when he serendipitously rejuvenated the field of parasite virology. Along with longtime collaborator Nicolas Fasel, PhD, of the University of Lausanne in Switzerland, and other colleagues, Beverley discovered that Leishmania parasites infected with a virus – dubbed Leishmaniavirus – cause significantly worse disease than those without a virus. Other researchers later showed that viruses in related parasites such as Trichomonas, which causes vaginal infections, and potentially Cryptosporidium, which causes diarrhea, also may exacerbate disease.
Beverley talked about the nascent field of parasite virology and his newest paper, an evolutionary study that suggests that Leishmania’s viruses may have helped the parasite infect vertebrates. The paper was published the week of Dec. 25 in Proceedings of the National Academy of Sciences.
What are parasite viruses?
It’s long been known that parasites, like every other creature, have their share of viruses. But for a long time, nobody was interested in them. Very few were infectious, they had never been associated with any diseases, and they were more or less dismissed as curiosities. But six or seven years ago, in the process of gathering evidence to answer a different question, we — along with Fasel’s lab — uncovered some really nice evidence that showed that Leishmaniavirus has a role in disease. If a Leishmania parasite is carrying this virus, there’s more severe disease, higher numbers of parasites, and the infection is more likely to metastasize. This is not what we were aiming to study, but you follow where the data goes. The data took us here, and off we went.
How do parasite viruses worsen parasitic diseases?
It’s important to remember that people are not infected with both a parasite and a virus. Rather, people are infected with a parasite that has a virus hidden inside it. What we think happens is that immune cells kill some invading parasites, releasing the virus. The body then generates an immune response to fight off the virus. But when you’re infected with a parasite, this is the wrong kind of immune response. It doesn’t kill the parasite; it seems to help the parasite make the disease worse.
Could you reduce parasitic disease by getting rid of the virus?
We think so, and we’re trying to do that in two ways. One, we know that drug treatment is more likely to fail in people infected with Leishmania that is carrying a virus. Since the virus increases parasite numbers, we think that such a scenario would call for a higher dose to kill them all. So we are talking with workers in countries where the parasite is endemic to develop a screening program to identify people infected with Leishmania parasites that are carrying Leishmaniavirus. They could then be treated with a higher dose of medicine. The other thing we’re trying to do is cure the parasite of the virus. We’ve found some antiviral compounds that work great in a culture dish, and we’re evaluating whether they can get rid of virus that is inside parasites that are inside animals — and, potentially, that are inside people.
What did you learn from surveying the evolutionary history of parasite viruses?
Leishmania is descended from parasites that infect insects, and only late in evolution did it learn how to infect vertebrates and then humans, and become a problem for us. This study started as a survey of a family of insect parasites related to Leishmania, which we performed in collaboration with a team led by Vyacheslav Yurchenko in the Czech Republic. But then we questioned whether these insect parasites could carry viruses. A lot of researchers are doing amazing work in viral discovery, but very few have hunted around in these oddball parasites. When we did, we found many new viruses, several of which were quite unique. But just as interesting is what we didn’t find – specifically, relatives of Leishmaniavirus in more ancestral lineages. This suggests that Leishmania acquired Leishmaniavirus about the same time it learned to parasitize vertebrates.
We were excited to find that one viral group common in the insect parasites included viruses distantly related to bunyaviruses, which are often animal pathogens. We named the new parasite virus that invaded the insect parasites “Leishbunyavirus,” or LBVs. While the LBVs that infected parasites within insects were quite similar to other bunyaviruses, in ongoing work we have found that the LBVs in Leishmania have undergone big changes that seem again to be related to increased virulence, via a mechanism completely different than in the original Leishmaniavirus.
So we have two lines of evidence that suggest something happened with the viruses at the same time that the parasite made the jump from insects to vertebrates. It’s very difficult to prove a role for viruses in disease by studying evolution, but the two viral “jumps” we have discovered provide a great corroboration of our experimental work, which shows that these viruses contributed to increased Leishmania parasite survival and disease pathology in vertebrates. Maybe without these viruses acting deep in evolution, these diseases would have become far less severe and terrible than they are now.







