Testing branched devices to fix iliac aneurysms
Washington University School of Medicine is part of a national trial to test a new device for fixing iliac aneurysms without open surgery
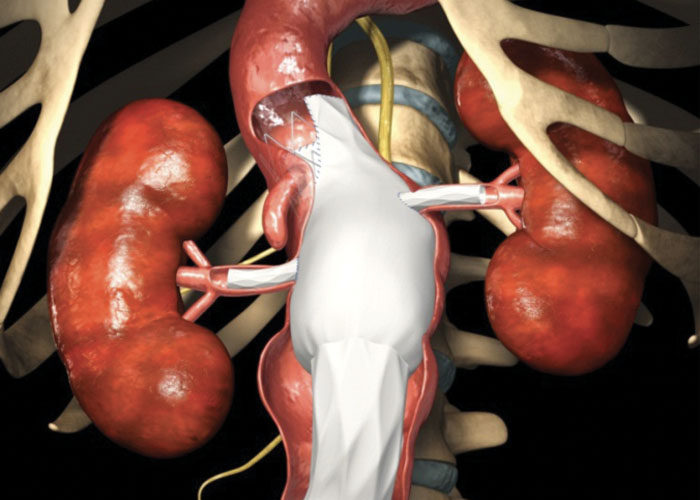
A clinical trial of an "off-the-shelf" fenestrated stent graft could broaden the ability of vascular surgeons to treat patients with complex aneurysms.
As leaders in vascular care, Washington University surgeons at Barnes-Jewish Hospital are participating in a clinical trial testing branched graft devices to fix iliac aneurysms, enlargements of the iliac arteries located in the pelvis. The branches allow surgeons to protect blood flow to the adjacent internal iliac artery that is often blocked by traditional grafts.
Some patients can handle this blockage if the collateral internal iliac artery remains open. But others may have low blood flow due to diseased internal iliac arteries on both sides. Without branched devices, these patients require open surgery to repair the aneurysm without occluding this artery.
“We would really like to be able to preserve both of the internal iliac arteries without open surgery,” said Luis Sanchez, MD, the Gregorio A. Sicard Distinguished Professor of Vascular Surgery and chief of vascular surgery at Barnes-Jewish Hospital. “These new devices add a small branch to a standard endovascular graft that we already use. If we need to go that far down on the reconstruction, we can still maintain that important internal iliac artery that goes into the pelvis.”
Washington University is among a select number of medical centers participating in the PRESERVE-Zenith Iliac Branch System Clinical Study to test the safety and efficacy of these devices.







