New way viruses trigger autoimmunity discovered
In mice, roseolovirus disrupts immune cells’ process of learning self-recognition
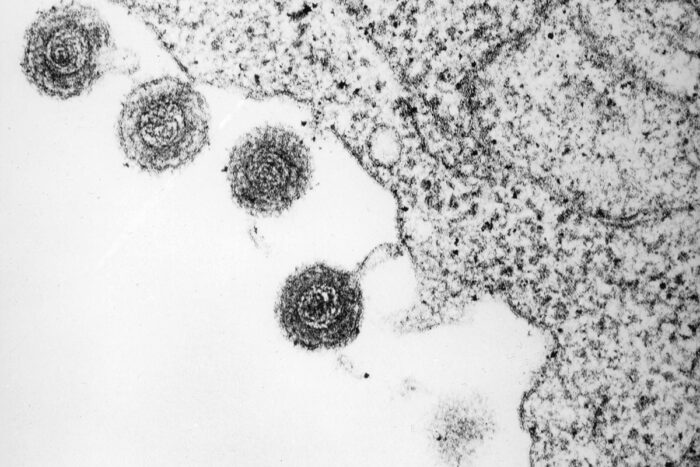 Bernard Kramarsky
Bernard KramarskyRoseolovirus particles emerge from an infected immune cell (above). Studying mice, researchers at Washington University School of Medicine in St. Louis have discovered that roseolovirus can trigger autoimmunity in a previously unknown way: by disrupting the process by which immune cells learn to avoid targeting their own body's cells and tissues.
Autoimmune diseases such as rheumatoid arthritis and Type 1 diabetes are thought to arise when people with a genetic susceptibility to autoimmunity encounter something in the environment that triggers their immune systems to attack their own bodies. Scientists have made progress in identifying genetic factors that put people at risk, but the environmental triggers have proven more elusive.
Researchers at Washington University School of Medicine in St. Louis have discovered that a viral infection can set a destructive process in motion, culminating in autoimmunity long after the infection has resolved. The researchers investigated the impact of viral infection on T cells, a group of immune cells that play a key role in many autoimmune conditions. In the study, which was conducted in mice, the researchers showed that murine roseolovirus infects the thymus — the organ where self-destructive T cells are identified and eliminated — and disrupts the screening process in the organ. Months after infection, the mice develop an autoimmune disease of the stomach driven by self-destructive T cells.
The study, published Feb. 28 in the Journal of Experimental Medicine, describes a previously unknown way a virus can trigger autoimmunity. Further, it suggests that human roseoloviruses, close relatives of murine roseolovirus, warrant investigation as possible causes of autoimmunity in people.
“It is very hard to find the culprit of a crime that was never even at the scene of the crime,” said senior author Wayne M. Yokoyama, MD, the Sam J. Levin and Audrey Loew Levin Professor of Arthritis Research. “As clinicians, we often look directly in the diseased tissue, and if we find no virus we conclude that the disease was not caused by a virus. But here we have a situation in which a virus is doing its damage someplace else entirely. This virus goes to the thymus, which is where T cells undergo a process to select those cells useful for immune defense but also get rid of T cells that are too likely to damage the body’s own tissues. And what we find is that this whole process, which is called central tolerance, is affected. T cells that shouldn’t leave the thymus get out, and they manifest months later in the stomach, causing an autoimmune disease in a location that was never infected with the virus.”
Human and mouse roseoloviruses are members of the herpesvirus family. In people, roseoloviruses cause roseola, a mild childhood illness that involves a few days of fever and rash. Most people have been infected with at least one roseolovirus by the time they start kindergarten. Like other herpesviruses, roseoloviruses cause lifelong infections, although the virus goes dormant and rarely causes symptoms after the initial infection.
Scientists have long suspected that roseoloviruses may be linked to autoimmunity. But the ubiquity of the viruses makes investigating any such connection difficult. It is hard to look for differences between infected and uninfected people when nearly everyone is infected early in life.
Instead, Yokoyama, first author Tarin Bigley, MD, PhD, a fellow in pediatric rheumatology, and colleagues studied murine roseolovirus, a recently discovered virus that infects the thymus and T cells of mice in the wild. The researchers infected newborn mice with the virus. Twelve weeks later, all of the mice had developed autoimmune gastritis, or stomach inflammation, though there were no signs of the virus in their stomachs. If the virus was promptly eliminated with antiviral drug treatment in the first few days, while it was still actively replicating, the mice did not develop gastritis three months later. If, however, the researchers waited to give an antiviral until the mice were 8 weeks old — after the active infection had resolved but before the mice showed signs of stomach problems — the drug did no good at all; the mice still went on to develop gastritis a few weeks later.
Scientists already knew that viral infection can lead to autoimmunity if some of the virus’s proteins happen to resemble normal human proteins. Antibodies meant to target the virus end up also reacting with normal human cells. The researchers found that the mice with gastritis had developed antibodies against proteins on stomach cells. But they also had developed antibodies against a wide array of normal proteins associated with other autoimmune conditions. In addition, they had many T cells that targeted the body’s own normal proteins, and other changes to the T cell population that biased the immune system toward autoimmunity.
“We don’t think the autoimmune gastritis is the result of molecular mimicry because we observed such a broad autoantibody response,” Bigley said. “The observation that infected mice produced diverse autoantibodies, in addition to the anti-stomach autoantibodies, suggested that murine roseolovirus infection early in life was inducing a wide-ranging defect in the body’s ability to avoid targeting its own proteins. This is why we focused our studies on the impact of infection on central tolerance rather than molecular mimicry.”
The next step is to investigate whether a similar process occurs in people.
“Human autoimmune disease also may occur via viral infection that gets cleared but leaves damage that can cause autoimmunity,” Yokoyama said. “But if so, there has to be some other factor that we don’t understand yet that makes some people more susceptible to the autoimmune effects of roseolovirus infection, because almost all people are infected, but most people do not get autoimmune diseases. That is a really important topic for further investigation.”







