New study shows breast tumors evolve in response to hormone therapy
Analysis of single tumor sample inadequate to provide best treatment
 M.J. Ellis
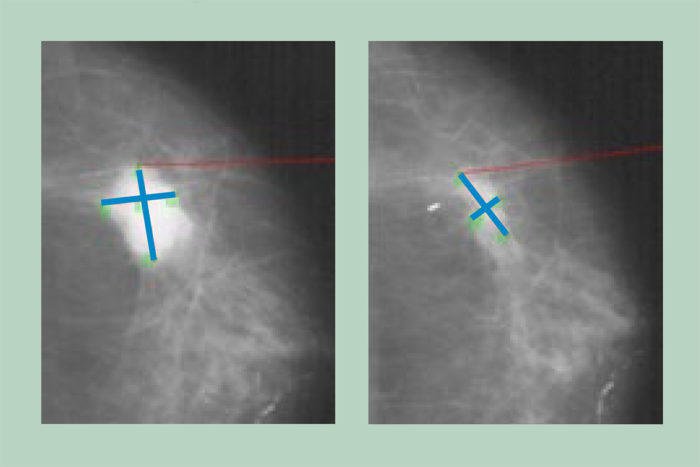
M.J. EllisThe scans show an estrogen-receptor-positive breast tumor before (left) and after four months of aromatase inhibitor therapy. This tumor was sensitive to aromatase inhibitors, shrinking in response to the lower estrogen levels produced by these drugs. A new study demonstrates that the genetics of these tumors can change dramatically in response to estrogen deprivation therapy, suggesting tumors should be analyzed more than once during treatment to assess whether doctors need to change therapeutic strategies.
Many breast tumors grow in response to female hormones, especially estrogen. Drugs that reduce estrogen levels in the body often are effective in reducing tumor size and preventing recurrence of the cancer. But some tumors become resistant to these therapies and continue to grow and spread.
A new analysis of breast tumors, before and after hormone-reduction therapy, reveals the extreme genetic complexity of these tumors and the variety of responses that are possible to estrogen-deprivation treatments. The findings also suggest that analyzing a single sample of the breast tumor is insufficient for understanding how a patient should best be treated.
The study, led by researchers at The McDonnell Genome Institute at Washington University School of Medicine in St. Louis and Baylor College of Medicine, appears Aug. 9 in the journal Nature Communications.
“Estrogen-receptor-positive breast cancers are not created equal,” said co-senior author Elaine R. Mardis, PhD, the Robert E. and Louise F. Dunn Distinguished Professor of Medicine and co-director of the McDonnell Genome Institute. “Each woman’s disease can have a range of responses to estrogen-lowering drugs. This study demonstrates that reducing estrogen levels in estrogen-receptor-positive breast cancer changes the genetics of the tumor, and these changes may be important for deciding how best to treat a patient after the surgical removal of the tumor.”
The researchers analyzed 22 breast tumors before and after four months of treatment with aromatase inhibitors, drugs commonly given to post-menopausal women with breast cancer. After menopause, the ovaries no longer produce estrogen, and aromatase inhibitors block the body’s remaining production of the hormone. Successful treatment reduces the size of a tumor before it is surgically removed, and the therapy has been shown to improve long-term outcomes for patients.
“In the post-treatment tumor samples, we found many new mutations or enrichment of mutations already seen in the pre-treatment samples,” said co-senior author Matthew J. Ellis, professor and director of the Lester and Sue Smith Breast Center at Baylor. “This means that under the environmental stress of the treatment, the tumors are spawning new sub-clones that subsequently can survive and grow despite therapy, and that is why we are having difficulty in the end treating estrogen-receptor-positive breast cancer. We found this result in the majority of tumors we studied.”
The majority of the tumors analyzed — 18 of 22 — had complex genetic landscapes and dynamic responses to hormone deprivation therapy, meaning that many of the gene mutations present in the tumors before and after treatment were different. For example, in one patient, certain mutations present in 92 percent of the initial tumor were totally absent in samples taken after four months of aromatase inhibitor therapy.
“The broad implication is that patients who undergo aromatase inhibitor therapy for several months prior to surgery should be re-evaluated immediately before their operation to determine how the tumor may have changed in response to the therapy,” Mardis said. “Such information can help indicate whether further estrogen suppression treatment is likely to contribute to a lower risk of relapse.”
The researchers analyzed only one tumor that had a complex but stable genetic landscape, meaning it was largely unchanged by aromatase inhibitor treatment. Another tumor had very simple and stable genetics before and after treatment. And two patient samples indicated evidence of two independent but intertwining tumors with separate genetic origins.
“It was surprising to find two ‘collision’ tumors in a group of only 22 patients,” said first author Christopher A. Miller, PhD, an instructor in medicine at Washington University. “This hints that collision tumors may be more common than we have previously realized. In these cases, estrogen suppression was the right approach for one of the tumors, but not the other, which limited the effectiveness of the treatment.”
“Our study also demonstrated that even single tumors can evolve in response to therapy very quickly,” Miller added. “This suggests that sequencing a tumor at diagnosis is not enough. Periodically scanning a tumor’s genome to understand how it is changing may ultimately help us evolve our treatment strategies to match.”
The study also reinforced past research suggesting that mutations in a gene called ESR1 are associated with resistance to aromatase inhibitor therapy, but the analysis did not identify any new genes that may also be responsible for conferring resistance to these drugs.







