Multifaceted research targets Wolfram syndrome, diabetes
Research at Washington University is looking at the causes and potential therapies for Wolfram syndrome, a rare genetic disorder related to diabetes
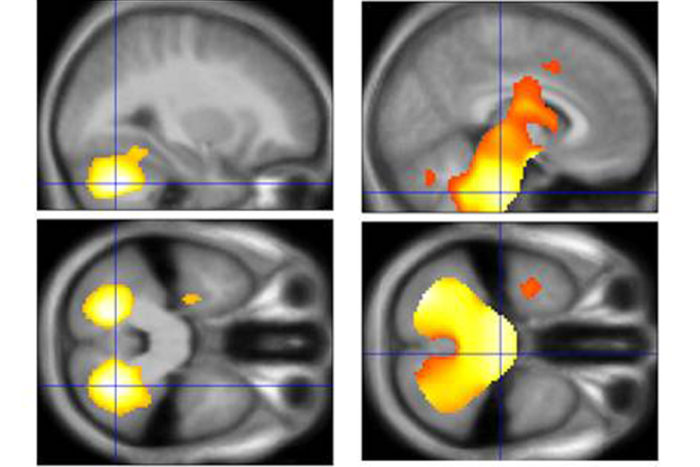
Brain scans identified regions of significantly reduced gray matter (right column) and white matter (left column) in young people with Wolfram syndrome.
Wolfram syndrome is a rare, genetic disease that begins with insulin-dependent diabetes and progresses to vision loss, kidney problems, deafness and serious neurological problems later in life. More than half of Wolfram patients die before their 40th birthday.
The gene that causes the disorder (WFS-1) was first discovered at Washington University School of Medicine in the 1990s by the late M. Alan Permutt, MD. Since that discovery, researchers and clinicians at Washington University have been at the forefront of the effort to understand what goes wrong in Wolfram syndrome and to develop therapies. Their work also may benefit those with less severe forms of diabetes.
International patient registry
In 2009, researchers at Washington University put together an international Wolfram syndrome registry to obtain genetic and clinical information about Wolfram patients. The following year they hosted the world’s first multidisciplinary research clinic for Wolfram syndrome patients, funded in part by the Snow Foundation, a family organization dedicated to raising funds for Wolfram syndrome research, and supported by an NIH grant.

As the effort has continued to grow, researchers Fumihiko Urano, MD, Bess Marshall, MD, and Tamara Hershey, PhD, have taken the lead on several basic and clinical research projects focused on the disorder.
Building on Permutt’s research, Urano is working to identify drug targets related to Wolfram syndrome. His lab also is looking for drugs that can relieve cell stress and potentially delay the progression of diabetes, vision loss and neurological problems in Wolfram syndrome. In addition, Urano is confident that studying this rare disease will assist scientists attempting to better understand more common forms of diabetes.
Applications for diabetes
“The diabetes in Wolfram syndrome is caused by interactions between genetic factors and a particular type of cell stress,” Urano said. “And the same type of cellular stress is involved in juvenile-onset diabetes (type 1) and adult-onset diabetes (type 2). The causes may be different, but a common biological process is altered in all three.”
Urano also is looking at potential treatments for the disorder, studying drugs that might slow the progress of the disease and looking at potential therapies that could replace or repair tissues that have been damaged by Wolfram syndrome.
“We have several candidate drugs, and we are looking for simple ways to asses how effective those drugs will be by looking at their actions in blood taken from patients,” Urano said. “At the same time, we are planning a clinical trial with Tamara Hershey and Bess Marshall that will help us learn how effective some drugs may be in treating the neurological features of Wolfram syndrome.”
Affects on the brain

By studying MRI scans conducted on the young people taking part in the annual Wolfram research clinics, Hershey and her colleagues have learned that the disease affects brain structure very early in its course.
“We know that the Wolfram gene is important throughout the body—in the heart, retina, pancreas and so on,” Hershey said. “The pancreas is affected very early and leads to diabetes, and our findings suggest the brain also is affected early in the disease.”
Hershey and her colleagues have detected changes in the size and volume of several brain structures in Wolfram patients. It appears that some parts of the brain continue to develop differently as the disease progresses.
“The neurological features of the disease may be the most feasible thing to target and monitor in clinical trials,” she explains. “That’s because by the time a child gets a diagnosis of Wolfram syndrome, the insulin-producing cells in the pancreas already are damaged or destroyed, and the child has developed insulin-dependent diabetes. By identifying time points at which it’s possible to intervene, we may be able to prevent some of the severe neurological problems that occur later in the course of Wolfram syndrome.”
Stem cells and damaged tissue
Meanwhile, Urano’s team is focusing on what to do if Wolfram syndrome is detected, looking for ways to replace damaged tissues by using a patient’s own cells. To accomplish this goal, Urano’s group has used skin cells taken from patients to create induced pluripotent stem cells (iPS cells).
“These iPS cells are a type of stem cell that can be generated directly from patients’ own tissue, including skin cells,” he said. “We can make pancreatic beta cells, brain cells and eye cells from these iPS cells. And we expect that these cells may one day be a cornerstone for the development of more effective treatments for Wolfram syndrome.”

Urano explained that the iPS cells first will be useful in identifying drugs to treat Wolfram syndrome and other diseases related to cell stress. In the future, he expects the cells also will be useful in regenerating damaged tissues, including cells in the pancreas that secrete insulin, retinal cells that are key to vision and even nerve cells.
In addition to the research efforts, Marshall is directing a clinical center of excellence for Wolfram syndrome patients through the Department of Pediatrics.
The clinicians at Washington University and St. Louis Children’s Hospital are now among the most experienced in the world at working with Wolfram patients, positioning the medical center as a unique resource for high quality interdisciplinary care for these unique and special patients.







