Malignant brain tumor care advances with greater precision and customization
New treatments and techniques are improving outcomes for patients with malignant brain tumors
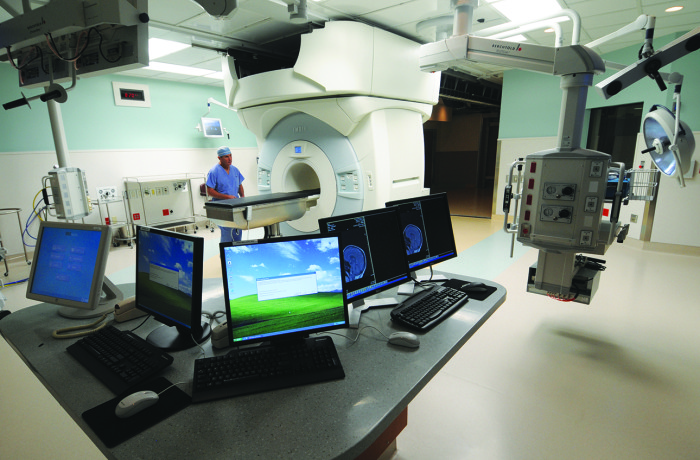
Room with iMRI equipment, which is used to help improve brain surgeries.
Malignant brain tumors are among the most challenging cancers to treat, but at the Alvin J. Siteman Cancer Center at Barnes-Jewish Hospital and Washington University School of Medicine, sophisticated imaging techniques and novel therapeutic approaches are boosting progression-free survival with fewer side effects. Over the past four years, Siteman has rounded out its spectrum of neuro-oncology care by adding a team of medical oncologists with special expertise in neurology.
“With the growth of our medical oncology program, we now offer comprehensive care for all brain malignancies,” says Mary Spencer, executive director of neuroscience and orthopedic centers at Barnes-Jewish Hospital.
Brain tumors are difficult to treat because surgeons can’t always remove the entire tumor without permanently damaging healthy brain tissue, and some tumors are inoperable because of where they’re located. Radiation therapy can access more remote areas, but like surgery, can also damage healthy tissue. Making matters worse, chemotherapies that could effectively target malignant tumors can’t cross the protective blood-brain barrier.
These challenges play a big part in how cancer patients feel about their diagnosis. “Patients worry about how the treatment will impact their quality of life. Their concerns shape how they perceive treatment options,” says radiation oncologist Christina Tsien, MD, division chief of neuro-radiation oncology. “These are very difficult decisions. At the Siteman Cancer Center, it takes a dedicated, multidisciplinary team to give patients the very best advice and care.”
Imaging enhances neurosurgery precision
Treatment for brain tumors begins with neurosurgeons. Those at Washington University and Barnes-Jewish Hospital have been early adopters of many of today’s most sophisticated techniques, says Ralph Dacey, MD, chair of neurological surgery.

Credit: Robert Boston.
Pre-surgical brain mapping uses functional MRI (fMRI) scanning to perform the critical job of locating tumors and critical healthy brain regions, but this can require hours of repeated scanning. Neurosurgeon Eric Leuthardt, MD, and neuroradiology pioneer Marcus Raichle, MD, recently developed a new form of fMRI mapping that takes only 15 minutes.
During surgery, intraoperative magnetic resonance imaging (iMRI) provides immediate, detailed feedback to assess progress and allow neurosurgeons to remove additional tumor on the spot. Clinical trials by neurosurgeon Michael Chicoine, MD, show that iMRI reduces the need for follow-up surgeries.
“It’s an amazingly powerful technology that allows us to remove tumors more completely the first time,” says Dacey. “We are now one of the most experienced teams in the country in using iMRI.”
For actual tumor removal, Leuthardt and Dacey are among the earliest to apply MRI-guided laser ablation technology to reach deep tumors; a precise, high-intensity laser inserted through a tiny hole in the skull eliminates tumor tissue as MRI assesses progress in real time.
“This tool gives us a treatment option for patients with tumors that were previously deemed inoperable and for patients who are not candidates for open surgery,” says Leuthardt. “It offers hope to certain patients who had few or no options before.”
Fine-tuning radiation oncology
Radiation oncology at Washington University School of Medicine and Barnes-Jewish Hospital has long been on the cutting edge of new technology. Siteman’s S. Lee Kling Proton Therapy Center is home to the world’s first compact proton beam accelerator, which Washington University physicians and scientists helped to develop and evaluate. Unique in the State of Missouri, its technology allows radiation oncologists to control the depth, shape and dose of radiation beams with great precision. The department was also the first in Missouri to offer the Gamma Knife, which delivers highly targeted radiation therapy from multiple angles.
In recent years, radiation therapy has become unbelievably precise, but this precision is of no use if it is not targeting the entire tumor. Part of the difficulty of treating brain tumors is their ability to infiltrate surrounding areas. It’s important to make sure that the most aggressive areas of tumor are treated and to limit the amount of normal tissue treated. The other difficulty is figuring out in real time whether a particular treatment is actually working effectively.
Recent breakthroughs in imaging can provide some of the necessary information regarding tumor structure to allow oncologists to better assess treatment response, improve radiation treatment planning and reduce the long-term toxicity of radiation to the normal brain. Tsien is leading an effort to validate some of these advanced imaging techniques in a large national study in newly diagnosed patients with glioblastoma, or GBM.
Minimizing damage to healthy brain is especially critical for pediatric and young adult patients because their brains are still developing, says Jeff Michalski, MD, vice chair of radiation oncology and chief of genitourinary services. Michalski is leading a major clinical trial of medulloblastoma, the most common pediatric brain cancer. The trial aims to reduce the dose and volume of radiation therapy.
Chemotherapy: Crossing the blood-brain barrier
One of the largest remaining challenges in treating brain malignancies is finding a way to carry chemotherapy across the blood-brain barrier. Many current chemotherapy agents have proven effective against brain malignancies, if only the treatments could reach the tumors. The neuro-oncology team has found a surprising way to make this happen; they discovered that the MRI laser ablation system they use to destroy tumor tissue also makes the treated area accessible to chemotherapy and possibly immune cells. The team is now enrolling patients in clinical trials to test whether combining laser ablation with chemotherapy or immunotherapy will increase survival. Early results are encouraging, says medical neuro-oncologist David Tran, MD, PhD, also a co-leader of the neuro-oncology research focus program.
“Laser ablation will destroy the tumor but it won’t destroy microscopic cancer cells in the surrounding area. If we can disrupt the blood brain barrier with laser ablation within this area, we can deliver chemotherapy and allow immune cells full access to kill residual cells.”
Tran’s team is pioneering a combination of this technology with novel immunotherapy drugs to achieve therapeutic synergy. His team is also testing personalized vaccines on GBM. Neurosurgeons can’t remove GBM because the tumor sprouts long tentacles that reach far into surrounding brain tissue. In the vaccine trials, cells from each patient’s brain tumor are combined with the patient’s immune cells and then reintroduced back into the patient in the hopes the sensitized immune cells will destroy the remaining tumor cells.
With these combined efforts, the neuro-oncology program aims to provide comprehensive and innovative care, delivered seamlessly, says Robert Portwood, MHA, director of operations for oncology services. That includes assisting patients with travel, scheduling appointments, finances, nutritional advice, and, if need be, planning end-of-life care. “Our goal is to not only offer premier care, but to provide our patients with all the services they need to have the best possible care experience,” says Portwood.







