Cancer genetic test offshoot helps diagnose rare syndrome in children
A genetic test developed from a tumor gene panel is helping to diagnose rare pediatric overgrowth syndromes
 Robert Boston
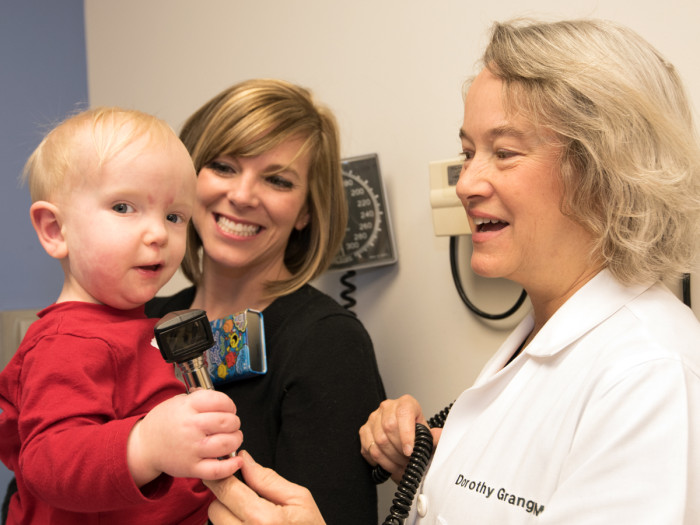
Robert BostonBaby Luke being examined by geneticist Dorothy K. Grange, MD, (right) while mother Stephanie watches. Luke has a rare childhood syndrome known as MCAP that causes overgrowth in certain parts of his body.
When Lucas was born, the doctor told Stephanie and Jason that their son had some ‘interesting characteristics.’ Baby Luke had a purple lace-like pattern over his entire body, his fingers and toes were webbed, one side of his body was larger than the other and he had a facial birthmark that concerned the doctor, who thought it might be associated with a neurological or blood vessel disorder.
After Stephanie and Jason searched the Internet for information on the characteristics, they could tell they were dealing with some type of genetic syndrome. To find out more, their doctor referred them to Washington University geneticist Dorothy K. Grange, MD.
“Pretty much as soon as I walked in the door I knew what he had,” says Grange. She had seen similar cases before and believed Luke had a rare condition called Megalencephaly-capillary malformation, or MCAP syndrome. It is also sometimes referred to as Macrocephaly-capillary malformation (M-CM).
“One of the first things Dr. Grange said to us was ‘Don’t Google this syndrome because it will give you worse case scenarios,’” says Stephanie. “Of course by that time we’d already done just that.”
A somatic mosaic syndrome
MCAP syndrome is a rare genetic disorder reported in only a few hundred children worldwide. Referred to as a somatic mosaic disorder, MCAP has complex genetic origins. Unlike an inherited disorder, which is passed on from a parent to a child, a somatic disorder begins after conception, as the embryo is forming. A genetic change, or mutation, arises early on in specific cells during the embryo’s development. Depending on where these cells develop, the mutation may end up in certain tissues of the body, but not others, creating a mosaic pattern.
The mutation leading to MCAP syndrome causes overgrowth of specific tissues in the body. In Luke’s case, the mutation caused overgrowth of his right side so that it appeared larger than his left side. He had overgrowth in certain small blood vessels in his face, which led to his birthmark. Some of his fingers and toes also had some webbing due to additional overgrowth in those tissues.
Another common hallmark of the MCAP syndrome is brain overgrowth. Patients like Luke require regular brain scanning to monitor for this overgrowth, which may lead to developmental delays and seizures.
From cancer to MCAP
To verify Luke’s clinical diagnosis, Grange wanted to perform a genetic test that would sequence the gene involved in the syndrome and pinpoint the overgrowth mutation. She knew Washington University’s Genomics and Pathology Services (GPS) had a somatic cancer genetic test that sequenced specific tumor types and included the same gene that was also involved in MCAP syndrome. The gene, called PIK3CA, is known to play a role in cell growth. It is mutated in several forms of cancer, as well as in MCAP syndrome and other overgrowth disorders. Even though the PIK3CA gene is affected in both tumor cells and in overgrowth disorders, there is fortunately no known increased risk for cancer in patients with syndromes like MCAP.

Grange asked GPS medical director, Catherine Cottrell, PhD, if she could help design a test for Luke based on the cancer panel. Cottrell and her colleagues agreed.
“Once we knew what gene was involved in the disorder, we could develop a test for it,” says Cottrell. “And because we were already sequencing tumors with similar gene mutations, we could apply the same technology to this new test.”
Just like a mutation that is found solely in a tumor and not in healthy cells, the MCAP mutation only occurs in certain tissues. So the challenge Grange faced was obtaining a sample from Luke that would contain enough of the mutation that it would show up on the test. Blood cells would likely not contain the mutation. So in addition to a blood sample, Grange took a buccal sample using a swab that lightly scrapes the inside of the mouth to gather cheek cells and the DNA they contain. The hope was that the cheek cells would contain more of the mutation they were looking for than the blood sample.
Now available to patients across the country, the overgrowth syndrome genetic test is known as the SOMA (for somatic) Gene Set, and can be ordered through a clinician. In Luke’s case the testing was covered by the family’s insurance. The test uses advanced DNA sequencing technology and analysis methods to characterize PIK3CA and a handful of other genes thought to be involved in certain overgrowth disorders. When Luke’s samples were sent for testing, 37% of the buccal swab cells and less than 1% of the cells in the blood sample contained the mutation in PIK3CA, confirming the MCAP syndrome diagnosis.
A relief to know
Once Grange received the results of the test from GPS, she sat down with Luke’s family and a genetic counselor to go over the findings.
“I was impressed by the report from the test – it was very thorough,” says Stephanie. “The whole time we were unsure about what Luke had,” adds Jason. “So it was a relief to finally know.”

Luke returns to St. Louis Children’s Hospital every few months for ultrasounds and scans to make sure he has no brain overgrowth. He has also had surgeries to fix a finger that was locked in a straightened position and to separate the fingers that had webbing between them. Doctors have checked his heart for any abnormalities and physical therapists monitor his growth, paying close attention to his right leg, which is longer than the left.
“I feel like we’re pretty fortunate,” says Stephanie. “Luke seems to be on the mild side of the syndrome compared to others I’ve connected with.”
While genetic testing is not new, sequencing technologies have advanced significantly in recent years and these tests are becoming increasingly common. “I use genetic testing in my practice every day,” says Grange. “I see children (and adults) with all kinds of genetic conditions.” Grange has a few other patients she sees who have now undergone the SOMA genetic test through GPS. The test has confirmed each one of their diagnoses.
“Having a molecular cause for the disorder could help clinicians decide on possible treatments and therapeutics as they continue to emerge,” says Cottrell. She adds that knowing the genetic cause of rare conditions like MCAP syndrome can solidify a diagnosis, help a physician understand how best to treat a patient, as well as provide genetic counseling to appropriate family members.








